Journal of Non Invasive Vascular Investigation Category: Clinical
Type: Opinion
Focus on: In the Treatment of AAA is there Yet a Space for Open Surgery?
*Corresponding Author(s):
Enrico RescignoDepartment Of Vascular Surgery, Leonardi And Riboli Hospital, Lavagna, Genoa, Italy
Tel:+39 3473574457,
Email:erescigno61@icloud.com
Received Date: Mar 13, 2017
Accepted Date: May 11, 2017
Published Date: Jun 02, 2017
Abstract
The Endo Vascular Aneurysm Repair (EVAR) has revolutionized the treatment of Abdominal Aortic Aneurysm (AAA) with low rate morbidity and mortality. A strong growth of EVAR with a progressive reduction of Open Repair (OR) is expected for the coming years. Despite this, there are important reasons in favour of open surgery: Patients unfit for EVAR with complex aortic anatomy, increasing risk of cancer related to computed tomography studies for endovascular graft surveillance, secondary interventions post EVAR, critically ill patients with ruptured - AAA not transferable to specialist vascular centres. In several cases AAA will still need open surgery and a new generation of vascular surgeons prepared for complex aortic anatomy will be required as well.
In the last decades, the evolution of Endo Vascular Aneurysm Repair has radically revolutionized the approach in vascular surgery of Abdominal Aortic Aneurysm with a reduced perioperative rate of mortality and adverse events [1]. Italian Vascular Society Registry - AAA shows over the time a progressive reduction of Open Repair from 1930 patients in 2008 to 887 in 2014 with a short term mortality of 1.1% for EVAR vs 6.2% for OR [2]. The availability of various devices with low risks (fenestrated devices, branched devices, parallel stent-grafts CHIPS: Chimney - Periscope-Sandwich, Off the shelf devices) and new investigational endovascular Aneurysm Sealing Systems (Neck angulation up to 90°, Inflatable Proximal Rings and Inflatable Endobag-Nelly System) have expanded EVAR indications. The forecast growth of EVAR for the coming years shows an increase in Italy from 62% in 2013 to 72% in 2023 [3]. Facing these results is there yet a space for open surgery (Figure 1).
In the last decades, the evolution of Endo Vascular Aneurysm Repair has radically revolutionized the approach in vascular surgery of Abdominal Aortic Aneurysm with a reduced perioperative rate of mortality and adverse events [1]. Italian Vascular Society Registry - AAA shows over the time a progressive reduction of Open Repair from 1930 patients in 2008 to 887 in 2014 with a short term mortality of 1.1% for EVAR vs 6.2% for OR [2]. The availability of various devices with low risks (fenestrated devices, branched devices, parallel stent-grafts CHIPS: Chimney - Periscope-Sandwich, Off the shelf devices) and new investigational endovascular Aneurysm Sealing Systems (Neck angulation up to 90°, Inflatable Proximal Rings and Inflatable Endobag-Nelly System) have expanded EVAR indications. The forecast growth of EVAR for the coming years shows an increase in Italy from 62% in 2013 to 72% in 2023 [3]. Facing these results is there yet a space for open surgery (Figure 1).
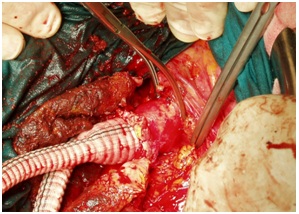
Figure 1: Infrarenal cross-clamp repair.
Keywords
Surgery
INTRODUCTION
Not suitable anatomy for EVAR: Aneurysm estension over renal arteries, absence of proximal aortic neck, presence of affixing thrombin in the proximity of aortic neck, extreme angles of abdominal aorta, stenotic and tortuose iliac artery, renal arteries abnormalities (Figure 2), colon ischemia (Figure 3) are anatomic conditions unfit for EVAR [4]. The new generation of devices may extend in the future the indications and overcoming unfit anatomy, so that more patients will be EVAR suitable and the remain open surgery will become increasingly complex. Usually complex anatomy requires suprarenal cross-clamp associated with higher rate of complications (eg: intraoperative blood loss, postoperative renal insufficiency, use of adjunctive renal and/or visceral grafts). Fewer surgeons will perform these repairs and fewer fellows will be able to complete the operation independently after training [5].
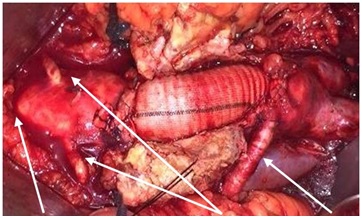
Figure 2: Retroaortic Left Renal vein/Renal arteries/Aberrant Renal artery.
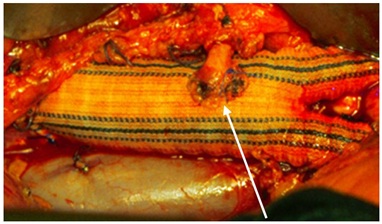
Figure 3: Inferior mesenteric artery reimplantation.
Younger patients without high risk for OR: A continued surveillance after EVAR to detect aneurysm growth, due to endoleak, device migration, or structural failure is recommended [6]. Computed Tomography (CT) studies are routinely used for graft surveillance in EVAR patients, but up to 2% of all cancers in US may be attributable to their radiations [7]. In the EVAR patients the risk of cancer is higher in CT surveillance vs alternative modalities and a higher risk is present in younger people, with highest in those aged 50 to 55 years and lowest in patients aged >/= 80 years [8].
Infected Aneurysm / Primary aortoenteric fistula: The gold standard of treatment for an infected aortic aneurysm is still surgical debridement of infected tissue, followed by in situ revascularization or extra-anatomic grafting and long-term antibiotic therapy. Endovascular repair should be an alternative for high risk patients or used as a bridge while they await an open procedure [9].
Connective tissue disease: The American Heart Association (AHA) guidelines say that stent grafts are controindicated in patients with Marfan Syndrome and related connective tissue disorders. Additionally, although stent grafting in these patients is feasible, post intervention surveillance confirms that the aorta continues to expand [10].
EVAR failure: Though the incidence is gradually declining, secondary interventions persist as the Achilles’ heel of EVAR. The rate of early conversion is about 1.5% (range 0.8%-5.9%) with an average mortality of 12.4%, the rate of late conversion is about 1.9% (range 0.4%-22%) with a mortality rate of 10%. A recent Cochrane review shows that EVAR mortality in short-term is significantly lower than OR, while in long-term there is no more difference [11]. A recent meta-analysis confirms the result in long term and argues that the mortality is related to AAA [12]. These data support the importance of both appropriate surveillance strategy and expert vascular surgeons in highly complex open interventions [13].
Unstable ruptured AAA (rAAA): Since the introduction of EVAR, there has been a significant decrease in the annual number of deaths related to treated rAAA from 44.3% in the period 1993-1998 to 39.9% in 2001-2005 [14]. A stratified comparison has demonstrated that in preoperative low and medium-risk rAAA patients, EVAR has a lower mortality and morbidity compared with OR, while in highest risk patients there was no difference [15]. OR remains, therefore, a viable option for high-risk patients and it becomes necessary for those patients afferent to a peripheral center without EVAR as our Hospital in Lavagna (Italy), organized on a HUB-SPOKE network for cardiovascular emergencies. The time necessary to transfers may erase the survival advantage guaranteed by HUB and therefore give rise the Hamletic question “to transfer or not to transfer”. In fact the survival advantage for transferred patients who receive EVAR, may be eclipsed by mortality of the transfer process up to 17% [16].
CONCLUSION
Although EVAR has revolutionized the AAA treatment, OR should not be left behind. Among all others (unfavourable anatomy, associated pathology, long-term results, treatment in emergency in the peripheral centers, etc) one of the reasons is also the need for OR after EVAR. For the increased number of EVAR procedures performed, is expected un increased as well for complex OR and therefore un appropriate education of young generation of vascular surgeons will be required.
REFERENCES
- Salzler GG, Meltzer AJ, Mao J, Isaacs A, Connolly PH, et al. (2015) Characterizing the evolution of perioperative outcomes and costs of endovascular abdominal aortic aneurysm repair. J Vasc Surg 62: 1134-1139.
- https://www.sicvereg.it/
- Millennium Research Group (2014) Millennium Research Group, Toronto, Canada.
- Bryce Y, Rogoff P, Romanelli D, Reichle R (2015) Endovascular repair of abdominal aortic aneurysms: vascular anatomy, device selection, procedure, and procedure-specific complications. Radiographics 35: 593-615.
- Landry G, Lau I, Liem T, Mitchell E, Moneta G (2009) Open abdominal aortic aneurysm repair in the endovascular era: effect of clamp site on outcomes. Arch Surg 144: 811-816.
- Chaikof EL, Brewster DC, Dalman RL, Makaroun MS, Illig KA, et al. (2009) The care of patients with an abdominal aortic aneurysm: the Society for Vascular Surgery practice guidelines. J Vasc Surg 50: 2-49.
- Brenner DJ, Hall EJ (2007) Computed tomography--an increasing source of radiation exposure. N Engl J Med 357: 2277-2284.
- Motaganahalli R, Martin A, Feliciano B, Murphy MP, Slaven J, et al. (2012) Estimating the risk of solid organ malignancy in patients undergoing routine computed tomography scans after endovascular aneurysm repair. J Vasc Surg 56: 929-937.
- Spelman D (2017) Overview of infected (mycotic) arterial aneurysm. UpTo Date, Wolters Kluwer, Netherlands.
- Hiratzka LF, Bakris GL, Beckman JA, Bersin RM, Carr VF, et al. (2010) 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM Guidelines for the Diagnosis and Management of Patients With Thoracic Aortic Disease: Executive Summary. Circulation 121: 1544-1579.
- Paravastu SC, Jayarajasingam R, Cottam R, Palfreyman SJ, Michaels JA, et al. (2014) Endovascular repair of abdominal aortic aneurysm. Cochrane Database Syst Rev CD004178.
- Powell JT, Sweeting MJ, Ulug P, Blankensteijn JD, Lederle FA, et al. (2017) Meta?analysis of Individual?patient Data From EVAR?1, DREAM, OVER and ACE Trials Comparing Outcomes of Endovascular or Open Repair for Abdominal Aortic Aneurysm Over 5 Years. Br J Surg 104: 166-178.
- Moulakakis KG, Dalainas I, Mylonas S, Giannakopoulos TG, Avgerinos ED, et al. (2010) Conversion to open repair after endografting for abdominal aortic aneurysm: a review of causes, incidence, results, and surgical techniques of reconstruction. J Endovsasc Ther 17: 694-702.
- Giles KA, Pomposelli F, Hamdan A, Wyers M, Jhaveri A, et al. (2009) Decrease in total aneurysm related deaths in the era of endovascular aneurysm repair. J Vasc Surg 49: 543-551.
- Ali MM, Flahive J, Schanzer A, Simons JP, Aiello FA, et al. (2015) In patients stratified by preoperative risk, endovascular repair of ruptured abdominal aortic aneurysms has a lower in-hospital mortality and morbidity than open repair. J Vasc Surg 61: 1399-1407.
- Mell MW, Wang NE, Morrison DE, Hernandez-Boussard T (2014) Interfacility transfer and mortality for patients with ruptured abdominal aortic aneurysm. J Vasc Surg 60: 553-557.
Citation: Rescigno E, Rosa G (2017) Focus on: In the Treatment of AAA is there Yet a Space for Open Surgery? J Non invasive Vasc Invest 2: 007.
Copyright: © 2017 Enrico Rescigno, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
© 2024, Copyrights Herald Scholarly Open Access. All Rights Reserved!