
Efficacy and Safety of Forced-Air Warming System versus Passive Warming Measures in Major Surgeries: A Systematic Review
*Corresponding Author(s):
Wenxi TangSchool Of International Pharmaceutical Business, China Pharmaceutical University, Nanjing, China, Center For Pharmacoeconomics And Outcomes Research, China Pharmaceutical University, Nanjing, China
Tel:+86 13770302713,
Email:tokammy@cpu.edu.cn
Abstract
Purpose: To compare the clinical impact of forced-air warming system (Bair HuggerTM, BH) and passive warming measures in major surgery patients.
Methods: Databases including Pubmed, Cochrane Library, Clinical Trials.Gov and CNKI were searched to collect studies published before January 2019 that were concerned the clinical effects of Bair Hugger. Two reviewers independently screened the literatures, extracted the data. The revised Jadad scale was used to evaluate the methodological quality of the literatures. Meta-analysis was performed by using Review Manager 5.3.0.
Findings: A total of 27 studies were included. The result of meta-analysis showed that BH had a significant advantage in rate of hypothermia (RD = -0.42, 95%CI (-0.68, -0.16)) , shivering (RD = -0.28, 95%CI (-0.43, -0.13)), anesthesia recovery time (MD = -8.27, 95% CI (-13.49, -3.05)), hospital stay (MD = -1.27, 95% CI (-2.05) , -0.48)), while incision infection RD = -0.15, 95%CI (-0.40,0.11)) , intraoperative blood loss (MD = -16.88, 95%CI(-34.73,0.96)), intraoperative blood transfusion (MD = -41.49, 95% CI( -108.36, 25.38)) , pain RD = -0.02, 95%CI(-0.08, 0.03)) and other complications (RD = -0.13, 95%CI(-0.39,0.12)) had an advantage but not significant. Subgroup analyses showed that operation mode and operation duration was the sensitive factors.
Conclusion: Compared to passive warming, Bair Hugger has significant advantages in hypothermia protection and further reduces the risk of incision chills and prolonged hospital stay. Combined with the current status of body temperature protection in China, it is necessary to enhance the awareness of body temperature protection, standardize medical behavior, and increase the popularity of active warming systems.
Keywords
Bair Hugger Forced-air warming; Hypothermia; Passive warming measure; Systematic review
List of Abbreviations
ASPAN: American Society of Peri-Anesthesia Nurses
BH: Bair Hugger™
CI: Confidence interval
CNKI: China National Knowledge Infrastructure
FAW: Forced-air warming system
NICE: National Institute for Health and Care Excellence
RD: Risk difference
MD: Mean difference
Introduction
Body temperature is an important vital sign of the human body. In normal conditions, body temperature is regulated by nerves-body fluids and maintained at approximately 37°C to ensure the stability of physiological functions [1]. However, during surgical operation, patients are prone to hypothermic events (core temperature < 36°C) due to the influence of various factors such as anesthesia, operating room temperature, and warming measure, intraoperative transfusion and infusion, and patients themselves, leading to intraoperative and postoperative rehabilitation risks [2,3]. In 2014-2015, Yi, et al., conducted a nationwide epidemiological survey in China, which showed that the incidence of intraoperative hypothermia was approximately 44.3% in this country [4].
The primary harm of intraoperative hypothermia is a resultant poor prognosis. Existing studies have shown that the occurrence of intraoperative hypothermia can affect blood coagulation function in patients and lead to increased bleeding amount and prolonged operation time; increase wound infection rate; slow down bodily and drug metabolism; impair the function of respiratory system; and increase the possibility of shivering and cardiovascular complications, causing a decline in patient satisfaction with treatment and quality of life [5,6],
Currently, the methods of intraoperative body temperature protection can be divided into passive warming and active warming. The former method achieves warming by increasing insulating material to reduce heat dissipation, such as covering a quilt, multilayer surgical drapes and insulating blanket. The latter method actively provides conductive, convective, or radiative heat energy, including warmed intravenous fluid and intraabdominal irrigation fluid for internal use, and circulating water mattress with alternating temperature and Forced-Air Warming System (FAW) for external use. It has gradually become a consensus in medical practice across various countries to apply prewarming before surgery and take active warming measures in a timely manner for patients presenting with preoperative or intraoperative hypothermia [4,7]. However, with regard to clinical practice, intraoperative body temperature protection is still primarily limited to passive warming for patients in China [2]. Yi, et al., showed that active warming devices were used in only 10.7% of Chinese patients, while the application of warming devices with higher efficiency has not yet been popularized [8].
Existing reviews on the effects of warming measures have mainly focused on the change of body temperature and the incidence of hypothermia, while lacking exploration into a series of adverse events caused by intraoperative hypothermia. In the present study, we selected the Bair Hugger™ (BH), a FAW blanket that performs well in active warming, to systematically evaluate its clinical effects versus passive warming measures. The results of this study could provide more sufficient clinical evidence for the selection of suitable warming method in the Perioperative period.
Methods
Search strategy
Computer searches were performed in the Pubmed, Cochrane Library, Clinical Trials.Gov, and China National Knowledge Infrastructure (CNKI) databases to retrieve studies related to the clinical effects of BH. The time limit of the search was set to January 2019. Search with “Bair Hugger” as the key word. The citations and grey literature were searched manually.
Inclusion and exclusion criteria
Inclusion criteria: The inclusion criteria were formulated according to the principle of PICOS. Target population: patients undergoing major surgery (with high complexity and high risk); intervention method: BH FAW blanket; control group: passive warming measures, such as covering a cotton blanket; outcome indicators: relevant clinical effects, such as infection and shivering; and study design: randomized clinical trial.
Exclusion criteria: 1. Articles that did not meet the inclusion criteria; 2. not published in Chinese or English; 3 incomplete outcome indicators, for example, not reporting the parameter estimation range; 4. Repeated publication; and 5. not intraoperative warming.
Literature selection, data extraction, and quality evaluation
Two researchers independently carried out literature selection, data extraction and quality evaluation. Divergence of opinions was resolved by discussion or assistance from a third party. Data extraction information included author, year of publication, country, sample size, basic patient information, type of surgery, intervention measures for the experimental group/control group, and outcome indicators. The quality of the included studies was evaluated using the modified Jadad scale [9,10]. The evaluation items comprised random sequence generation, randomization concealment, blinding method, withdrawal and dropout. The scoring criteria are listed in table 1. The quality of literature was indicated by the score: 1-3 points for low quality and 4-7 points for high quality.
|
|
Randomization |
Concealment of allocation |
Double blinding |
Withdrawals and dropouts |
Total |
|
Appropriate |
Random numbers, computer generated, etc |
Central allocation, sequentially numbered drug containers of identical appearance, etc |
Identical placebo or similar method was used |
Reasons and numbers were stated |
2 |
|
Unknow |
Insufficient information about the sequence generation process to permit judgement |
Insufficient information to permit judgement |
The trial was described as blind, but the method was not described |
Insufficient information to permit judgement |
1 |
|
Inappropriate |
Non-random component in the sequence generation process |
Participants could possibly foresee assignments and thus introduce selection bias |
No blinding or blind methods is inappropriate |
Reasons and numbers were not stated |
0 |
Table 1: Evaluation items and scoring criteria of literature quality.
Statistical analysis: The effect size of count data and measurement data was estimated using Risk Difference (RD) and Mean Difference (MD), respectively. The 2 test was used to evaluate the heterogeneity of included study results, and I2 was used to quantitatively determine the level of heterogeneity. If there was low statistical heterogeneity between the results of different studies, the fixed-effects model was used for meta-analysis; if there was high statistical heterogeneity, the source of heterogeneity was further analyzed, and if there was evident clinical heterogeneity, subgroup analysis or sensitivity analysis was performed, or only qualitative description was used; otherwise, the random-effects model was adopted for meta-analysis. For all tests the level of significance was set at α = 0.05.
Results
Literature retrieval results
A total of 289 relevant articles were retrieved through the preliminary search, and 27 articles were finally included after stepwise selection. The procedure of literature selection is shown in figure 1.
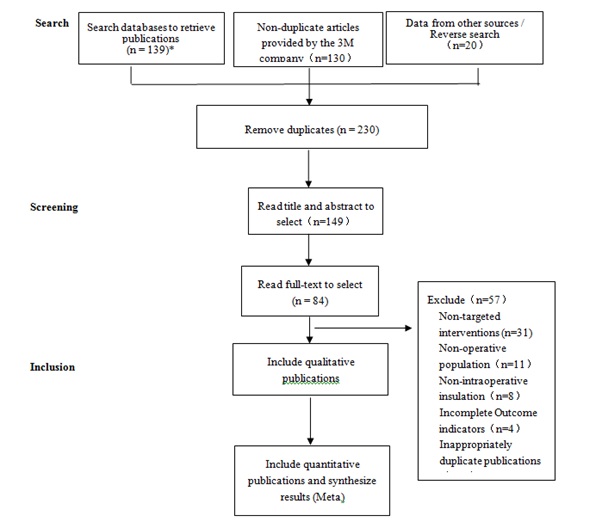
Figure 1: Flowchart of literature retrieval.
*English databases (PubMed, Cochrane Library, and Clinical Trials.Gov) = 126; Chinese database CNKI = 13
Basic characteristics of included studies
The basic characteristics of the included studies are summarized in table 2.
|
Studies |
Country |
Sample size |
Age |
Gender |
Surgery type |
Category |
Insulation measures for control group |
Length of surgery |
|
Pu Y 2013 [11] |
China |
55 |
68±11(44–89) |
27/28 |
laparoscopic gastrointestinal surgery |
laparoscopy |
no warming intervention |
1:146 ± 47 min |
|
55 |
67±11(43–83) |
33/22 |
2:149 ± 46 min |
|||||
|
Yi J 2018 [12] |
China |
30 |
57.9±11.8 |
21/9 |
open thoracic surgery and hip replacement surgery |
non-laparoscopy |
cotton blanket |
/ |
|
32 |
58.5±11.5 |
25/7 |
||||||
|
Takashi |
Japan |
20 |
61.8±2.5 |
9-Nov |
open abdominal surgery |
non-laparoscopy |
warming blankets |
168.8 ± 16.2 min |
|
Matsukawa 1994 [13] |
20 |
61.3±3.0 |
16/4 |
|||||
|
V.Pathi 1996 [14] |
England |
29 |
61.8±1.3 |
19/10 |
cardiac operations |
non-laparoscopy |
passive rewarming with an aluminum space blanket |
/ |
|
26 |
61.6±1.6 |
18/8 |
||||||
|
Debra S.Mason 1998 [15] |
America |
32 |
38.5±6.1 |
Feb-30 |
Roux-en-Y Gastric Bypass |
non-laparoscopy |
warming blankets |
1:156.1 ± 27.4min |
|
32 |
40.7±9.6 |
25-Jul |
2:156.9 ± 31.6min |
|||||
|
R. Lindwall 1998 [16] |
Sweden |
12 |
65 ± 18 |
/ |
extensive thoracoabdominal operations under standard combined general and regional anaesthesia |
non-laparoscopy |
conservative passive heat preservation techniques |
1:280min |
|
13 |
66 ± 10 |
/ |
2:287min |
|||||
|
Marianne Winkler 2000 [17] |
Australia |
75 |
65 ± 11 |
37/38 |
hip arthroplasty |
non-laparoscopy |
conventionally warmed |
1:102 ± 36 min |
|
75 |
64 ± 10 |
28/47 |
2:97 ± 36 min |
|||||
|
Wei YR 2012 [18] |
China |
50 |
6.7 ± 5.5(d) |
68/32 |
surgery in neonates |
non-laparoscopy |
|
>2h |
|
25 |
7.2 ± 4.2(d) |
room temperature was adjusted by the 38°C open warm bed, the warm bed was not closed during the operation |
||||||
|
25 |
6.4 ± 5.2(d) |
room temperature was adjusted by the 38°C open warm bed, the warm bed was closed according to the heat tolerance |
||||||
|
V. N 2006 [19] |
China |
30 |
67.3 ± 9.1 |
21-Sep |
total knee replacement |
non-laparoscopy |
electric heating pad |
1:89.3±12.6 min |
|
30 |
67.4 ± 7.4 |
22-Aug |
2:90.9±13.8 min |
|||||
|
Alexander J. Butwick 2007 [20] |
America |
15 |
36 ± 2 |
0/30 |
cesarean delivery |
non-laparoscopy |
general cover |
1:41±10 min |
|
15 |
32 ± 6 |
2:52±17 min |
||||||
|
K. K. Leung 2007 [21] |
China |
30 |
66.1 ± 10.0 |
19/11 |
cesarean delivery |
non-laparoscopy |
electric heating pad |
1:271±113 min |
|
30 |
64.1 ± 12.0 |
20/10 |
2:258±148 min |
|||||
|
Jan L. De Witte 2010 [22] |
Belgium |
9 |
66 ±12 |
3-Jun |
colorectal surgery |
non-laparoscopy |
cotton blankets |
1:128±47min |
|
8 |
59 ±10 |
3-May |
2:114±42min |
|||||
|
Sung Hee Chung 2012 [23] |
Korea |
15 |
31.8 ± 3.9 |
0/45 |
cesarean section |
non-laparoscopy |
control group |
<2h |
|
15 |
31.9 ± 4.6 |
|||||||
|
Song RY 2016 [24] |
China |
35 |
≥ 60 |
27/8 |
thoracic surgery |
non-laparoscopy |
cotton blanket |
>2h |
|
35 |
20/15 |
|||||||
|
Wang YJ 2016 [25] |
China |
25 |
40-60 |
0/100 |
hysterectomy |
non-laparoscopy |
cotton blanket |
>2h |
|
25 |
40-60 |
|||||||
|
25 |
65-80 |
|||||||
|
25 |
40-60 |
|||||||
|
25 |
40-60 |
|||||||
|
25 |
65-80 |
|||||||
|
Liu C 2018 [26] |
China |
30 |
> 65 |
16/14 |
Joint replacement, open spine surgery |
non-laparoscopy |
cotton blanket |
1:150.17 ± 19.14min |
|
30 |
15/15 |
2:145.00 ± 24.87min |
||||||
|
Liang H 2016 [27] |
China |
29 |
57.54 ± 13.84 |
16-Dec |
hip arthroplasty |
non-laparoscopy |
conservative passive heat preservation techniques |
>2h |
|
35 |
53.94 ± 14.63 |
17/18 |
||||||
|
Ernst-Peter Horn 2002 [28] |
America |
15 |
33 ± 4 |
0/30 |
cesarean section |
non-laparoscopy |
|
1:38 ± 9min |
|
15 |
31 ± 5 |
intraoperative warming |
2:37 ± 12min |
|||||
|
Keun Man Shin 2015 [29] |
Korea |
36 |
56 ± 15 |
26-Oct |
endovascular coiling of cerebral aneurysms |
laparoscopy |
cotton blanket |
1:100 ± 45min |
|
36 |
60 ± 13 |
14/22 |
2:100 ± 37min |
|||||
|
Qu DC 2016 [30] |
China |
20 |
60-80 |
10-Oct |
total knee replacement |
non-laparoscopy |
cotton blanket |
1:89 ± 6.15min |
|
20 |
9-Nov |
2:90 ± 10.80min |
||||||
|
Siew-Fong Ng-2002 [31] |
Singapore |
100 |
66.27 ± 0.91 |
49/251 |
hip arthroplasty |
non-laparoscopy |
Two cotton blankets |
<2h |
|
100 |
66.61 ± 0.73 |
|||||||
|
Lai ZY 2017 [32] |
China |
25 |
68.3 |
15-Oct |
total knee replacement |
non-laparoscopy |
no warming intervention |
1:61.9 ± 14.2min |
|
25 |
67.8 |
13-Dec |
2:60.8±13.4min |
|||||
|
Isabelle Murat 1994 [33] |
France |
26 |
14.7 ± 1.7 |
/ |
spinal surgery |
non-laparoscopy |
surgical drapes |
1:5.8 ± 2.0h |
|
25 |
14.9 ± 2.2 |
|
2:4.8 ± 1.2h |
|||||
|
15 |
32 ± 6 |
|
|
|||||
|
Pei LJ 2018 [34] |
China |
50 |
54 ± 12 |
23/27 |
hip arthroplasty |
non-laparoscopy |
cotton gown and single layer of cloth surgical draping |
>2h |
|
50 |
57 ± 11 |
32/18 |
||||||
|
48 |
54 ± 12 |
24/24 |
||||||
|
48 |
50 ± 12 |
19/29 |
||||||
|
49 |
54 ± 13 |
25/24 |
||||||
|
47 |
57± 11 |
25/22 |
||||||
|
Gary M Onik 1993 [35] |
America |
44 |
/ |
/ |
Hepatic cryosurgery |
non-laparoscopy |
without Bair Hugger |
>2h |
|
28 |
/ |
/ |
||||||
|
Yuan GJ 2013 [36] |
China |
30 |
32-71 |
60/30 |
laparotomy |
non-laparoscopy |
|
>2h |
|
30 |
electric heating pad |
|||||||
|
30 |
electric heating pad and cotton blanket |
|||||||
|
Katie Hooven 2011 [37] |
America |
77 |
64.87 |
36/41 |
Colorectal surgery |
non-laparoscopy |
No warming |
<2h |
|
72 |
62.9 |
37/35 |
Table 2: Basic characteristics of included studies.
Quality evaluation of included studies
Among the 27 articles included, six (22.22%) were high-quality articles and 21 (77.78%) were low-quality articles. Ten of the low-quality articles scored 3 points (47.62%; Table 3). Overall, the quality of the included studies was not high.
|
Studies |
Randomization |
Concealment of allocation |
Withdrawals and dropouts |
Total |
|
Wei YR 2012 |
0 |
0 |
1 |
1 |
|
Pu Y 2013 |
1 |
1 |
1 |
3 |
|
Yi J 2018 |
2 |
1 |
1 |
4 |
|
Takashi Matsukawa 1994 |
0 |
0 |
1 |
1 |
|
V. Pathi 1996 |
1 |
1 |
1 |
3 |
|
Debra S. Mason 1998 |
1 |
1 |
1 |
3 |
|
R. Lindwall 1998 |
0 |
0 |
1 |
1 |
|
Marianne Winkler 2000 |
1 |
1 |
1 |
3 |
|
V. N 2006 |
0 |
0 |
1 |
1 |
|
Alexander J. Butwick 2007 |
1 |
1 |
1 |
3 |
|
K. K. Leung 2007 |
1 |
1 |
1 |
3 |
|
Jan L. De Witte 2010 |
2 |
2 |
1 |
5 |
|
Sung Hee Chung 2012 |
1 |
1 |
1 |
3 |
|
Song RY 2016 |
2 |
1 |
1 |
4 |
|
Wang YJ 2016 |
2 |
1 |
1 |
4 |
|
Liu C2018 |
0 |
0 |
1 |
1 |
|
Liang H 2016 |
1 |
1 |
1 |
3 |
|
Ernst-Peter Horn 2002 |
0 |
0 |
1 |
1 |
|
Keun Man Shin 2015 |
1 |
1 |
1 |
3 |
|
Qu DC 2016 |
2 |
1 |
1 |
4 |
|
Siew-Fong Ng 2002 |
2 |
2 |
1 |
5 |
|
Lai ZY 2017 |
0 |
0 |
1 |
1 |
|
Isabelle Murat 1994 |
1 |
1 |
1 |
3 |
|
Pei LJ 2018 |
1 |
0 |
1 |
2 |
|
Yuan GJ 2013 |
1 |
0 |
1 |
2 |
|
Gary M Onik 1993 |
0 |
0 |
0 |
0 |
|
Katie Hooven 2011 |
0 |
0 |
1 |
1 |
Table 3: Quality evaluation of included studies.
Meta-analysis results
Compared with the passive warming group, the patients using BH had lower incidence of hypothermia [RD = –0.42, 95% confidence interval (CI: –0.68, –0.16)], lower incidence of shivering [RD = –0.28, 95% CI (–0.43, –0.13)], shorter anesthesia recovery time [MD = –8.27, 95% CI (-13.49, -3.05)], and shorter hospital stay [MD = –1.27, 95% CI (–2.05, –0.48)]. The BH group also showed advantages in terms of other indicators including postoperative incision infection rate [RD = –0.15, 95% CI (–0.40, 0.11)], intraoperative blood volume [MD = –16.88, 95% CI (–34.73, 0.96)], intraoperative blood transfusion volume [MD = –41.49, 95% CI (–108.36, 25.38)], pain [RD = –0.02, 95% CI (–0.08, 0.03)], and other complications [RD = –0.13, 95% CI (–0.39, 0.12)], albeit not significant (Figure 2).
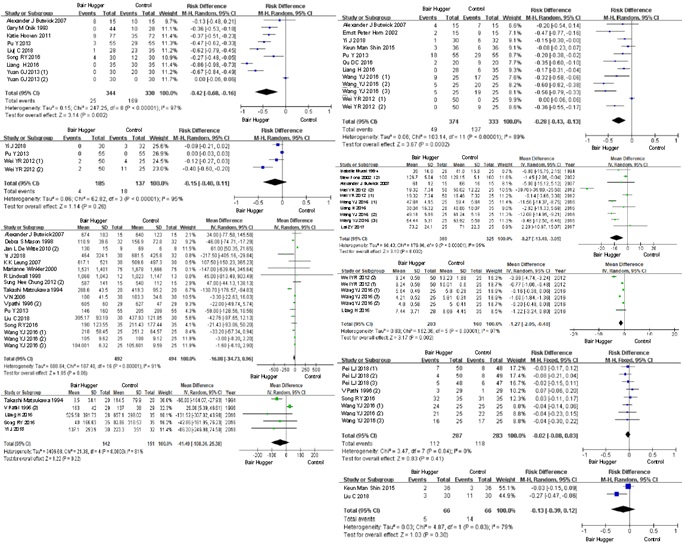 Figure 2: Meta-analysis results.
Figure 2: Meta-analysis results.
Note: On the left, from top to bottom are the results of incidence of hypothermia (%), incision infection rate (%), intraoperative blood loss volume (ml) and blood transfusion volume (ml); on the right, from top to bottom are the results of shivering (%), anesthesia recovery time (min), hospital stay (d), pain (%) and other complications (%).
Subgroup analysis results
To reduce the heterogeneity of the study results and explore the clinical effects of BH in more scenarios, subgroup analysis was carried out on the type of surgery (laparoscopy vs. non-laparoscopy), duration of surgery (with a 2-h boundary), and their combination. The heterogeneity was reduced, while the number of outcome indicators showing significant advantages in the BH group was increased. The number and heterogeneity of the included articles are listed in, and the results of the subgroup analysis are summarized in (Table 4 and 5).
|
|
laparoscopy |
non-laparoscopy |
<2h |
≥2h |
laparoscopy≥2h |
non-laparoscopy<2h |
non-laparoscopy≥2h |
|||||||
|
|
numbers |
I2 |
numbers |
I2 |
numbers |
I2 |
numbers |
I2 |
numbers |
I2 |
numbers |
I2 |
numbers |
I2 |
|
hypothermia RD, % |
1 |
/ |
7 |
97% |
5 |
98% |
3 |
70% |
1 |
/ |
5 |
96% |
2 |
85% |
|
incision infection rate RD, % |
1 |
/ |
2 |
77% |
/ |
/ |
2 |
97% |
1 |
/ |
/ |
/ |
1 |
80% |
|
intraoperative blood loss MD, ml |
1 |
/ |
14 |
92% |
5 |
88% |
8 |
81% |
1 |
/ |
5 |
88% |
7 |
82% |
|
blood transfusion MD, ml |
/ |
/ |
5 |
81% |
/ |
/ |
3 |
0% |
/ |
/ |
/ |
/ |
3 |
0% |
|
shivering RD, % |
2 |
7% |
7 |
92% |
4 |
56% |
4 |
94% |
1 |
/ |
3 |
0% |
3 |
96% |
|
anesthesia recovery time MD, min |
/ |
/ |
7 |
95% |
3 |
0% |
4 |
94% |
/ |
/ |
3 |
0% |
4 |
94% |
|
hospital stay MD, d |
/ |
/ |
3 |
97% |
/ |
/ |
3 |
97% |
/ |
/ |
/ |
/ |
3 |
97% |
|
pain RD, % |
/ |
/ |
4 |
0% |
/ |
/ |
3 |
0% |
/ |
/ |
/ |
/ |
3 |
0% |
|
other complications RD, % |
1 |
/ |
1 |
/ |
1 |
/ |
1 |
/ |
/ |
/ |
/ |
/ |
1 |
/ |
Table 4: Number and heterogeneity of articles included in the subgroup analysis.
|
|
Laparoscopy |
Non-laparoscopy |
<2h |
≥ 2h |
Laparoscopy ≥ 2h |
Non-laparoscopy <2h |
Non-laparoscopy ≥ 2h |
|
Hypothermia RD, % |
3/55 vs 29/55a |
-0.41 |
-0.4 |
-0.46 |
3/55 vs 29/55 a |
-0.3 |
-0.45 |
|
P<0.01 |
(-0.71 -0.12) |
(-0.77, -0.03 |
(-0.65, -0.28) |
P<0.01 |
(-0.60, 0.00) |
(-0.80, -0.10) |
|
|
Incision infection rate RD, % |
0/55 vs 0/55 a |
-0.19 |
/ |
-0.17 |
0/55 vs 0/55 a |
/ |
-0.25 |
|
(-0.37, -0.01) |
(-0.59, 0.26) |
(-0.54, 0.03) |
|||||
|
Intraoperative blood loss MD, ml |
146 vs 205 a |
-14.98 |
31.16 |
-26.43 |
146 vs 205 a |
31.16 |
-24.85 |
|
P=0.043 |
(-33.18, 3.21) |
(-17.83, 80.16) |
(-41.31, -11.55) |
P=0.043 |
(-17.83, 80.16) |
(-39.87, -9.83) |
|
|
Blood transfusion MD, ml |
/ |
-41.49 |
/ |
-120.04 |
/ |
/ |
-120.04 |
|
(-108.36, 25.38) |
(-161.62, -78.45) |
(-161.62, -78.45) |
|||||
|
Shivering RD, % |
-0.15 |
-0.31 |
-0.25 |
-0.31 |
18/55 vs 29/55 a |
-0.34 |
-0.33 |
|
(-0.28,-0.03) |
(-0.51, -0.11) |
(-0.44, -0.06) |
(-0.55, -0.06) |
P=0.041 |
(-0.51, -0.17) |
(-0.64, -0.02) |
|
|
Anesthesia recovery time MD, min |
/ |
-8.27 |
-1.47 |
-10.7 |
/ |
-1.47 |
-10.7 |
|
(-13.49, -3.05) |
(-2.86, -0.09) |
(-16.74, -4.65) |
(-2.86, -0.09) |
(-16.74, -4.65) |
|||
|
hospital stay MD, d |
/ |
-1.27 |
/ |
-1.27 |
/ |
/ |
-1.27 |
|
(-2.05, -0.48) |
(-2.05, -0.48) |
(-2.05, -0.48) |
|||||
|
pain RD, % |
/ |
-0.02 |
/ |
-0.03 |
/ |
/ |
-0.03 |
|
(-0.08, 0.03) |
(-0.09, 0.03) |
(-0.09, 0.03) |
|||||
|
other complications RD, % |
2/36 vs 3/36a |
3/30 vs 11/30a |
2/36 vs 3/36a |
3/30 vs 11/30 a |
/ |
/ |
3/30 vs 11/30 a |
|
P=1.000 |
P=0.046 |
P=1.000 |
P=0.046 |
P=0.046 |
Table 5: Subgroup analysis results.
Note: Bold text indicates a statistically significant difference. a: There was only one article.
Discussion and Conclusion
This study conducted a relatively comprehensive exploration into the indicators of the clinical effects of different warming measures. The results indicate that compared with passive warming measures, BH has remarkable advantages in reducing the risk related to hypothermia, shivering, and hospital stay in major surgeries. The results of the basic analysis are further supported by the subgroup analysis with more detailed subgroups.
In the case of high heterogeneity when combining some of the clinical evidence, it is impossible to perform subgroup discussions on the surgical site-a more critical factor-due to limited quantity of literature. Therefore, we conducted a subgroup analysis with regard to the characteristics of treatment regimen (laparoscopy/non-laparoscopy; surgical duration). We also performed a subgroup analysis based on the quality of literature. However, none of these analyses helped reduce the heterogeneity. This result might be related to factors such as the type of surgery and the year/country of publication that corresponded to the literature, and the long chain of evidence that led to the clinical results.
To date, intraoperative body temperature protection and monitoring have become a consensus in medical practice across various countries: Institutions such as the American Society of Peri-Anesthesia Nurses (ASPAN), National Institute for Health and Care Excellence (NICE), and Chinese Society of Anesthesia all recommend prewarming before surgery, continuous monitoring of body temperature and keeping the patient warm during surgery, and taking immediate active warming measures for patients with preoperative/intraoperative hypothermia [38]. However, the utilization rate of active warming devices in China is still not high, which would increase the risk of intraoperative hypothermia and other related events in patients, further causing a greater economic burden [8].
In summary, considering the difference of patient protection by various warming measures and the relatively low utilization rate of active warming devices in China, it is necessary to strengthen the awareness of patients and medical staff on body temperature protection, standardizes the medical behavior, and improve the application popularity of active warming measures.
Funding
This study was supported by project of Postgraduate Education Reform under “double first-class” capability construction from China Pharmaceutical University (Grant NO: 3151920118), provided to WT.
Author’s Contribution
WT conceptualized the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study design: AM, WT. Literature review: HX, YL, WT, XG. Data analysis: HX, XG, YM, YL. Drafting of the manuscript: HX, WT.
Acknowledgments
We thank Qiang Liu, the former advanced health economist engineer in Health Care Business Group Medical and Clinical Affairs, 3M China R&D Center, for providing some articles about BH.
References
- Hart SR, Bordes B, Hart J, Corsino D, Harmon D (2011) Unintended perioperative hypothermia. Ochsner J 11: 259-270.
- Ma ZL, Yi J (2017) Consensus among experts on prevention and treatment of perioperative patients with hypothermia. Medical Journal of Peking Union Medical College Hospital 8: 352-358.
- Burns SM, Kathy P, Guy C, Wojnakowski M (2010) Incidence of Postoperative Hypothermia and the Relationship to Clinical Variables. J Perianesth Nurs 25: 286-289.
- Yi J, Lei Y, Xu S, Si Y, Li S, et al. (2017) Intraoperative hypothermia and its clinical outcomes in patients undergoing general anesthesia: National study in China. PLoS One 12: 177221.
- Yu DN, Li D (2014) The harm and treatment of hypothermia. International Journal of Surgery 05: 258-261.
- Cheng ZN, Zhang J, Li X, et al. (2018) Intraoperative hypothermia in patients undergoing surgery. World Latest Medicine Information 18: 52-53.
- NICE Clinical Guidelines (2016) Hypothermia: prevention and management in adults having surgery. London: National Institute for Health and Clinical Excellence, UK.
- Yi J, Ziyong X, Xiaoming D, Fan T, Fu R, et al. (2015) Incidence of Inadvertent Intraoperative Hypothermia and Its Risk Factors in Patients Undergoing General Anesthesia in Beijing: A Prospective Regional Survey. PLoS One 10: 136136.
- Clark HD, Wells GA, Huet C, McAlister FA, Salmi LR, et al. (1999) Assessing the quality of randomized trials: reliability of the Jadad scale. Control Clin Trials 20: 448-452.
- Bhandari M, Richards RR, Sprague S, Schemitsch EH (2001) Quality in the reporting of randomized trials in surgery: is the Jadad scale reliable?. Control Clin Trials 22: 687-688.
- Pu Y, Cen G, Sun J, Gong J, Zhang Y, et al. (2014) Warming with an underbody warming system reduces intraoperative hypothermia in patients undergoing laparoscopic gastrointestinal surgery: a randomized controlled study. Int J Nurs Stud 51: 181-189.
- Yi J, H Liang, R Song, Xia H, Huang Y (2018) Maintaining intraoperative normothermia reduces blood loss in patients undergoing major operations: a pilot randomized controlled clinical trial. BMC Anesthesiol 18: 126.
- Matsukawa T, Kashimoto S, Nakamura T, Kume M, Kanda F, et al. (1994) Effects of a forced-air system (Bair Hugger, OR-type) on intraoperative temperature in patients with open abdominal surgery[J]. J Anesth 8: 25-27.
- Pathi V, Berg GA, Morrison J, Cramp G, McLaren D, et al. (1996) The benefits of active rewarming after cardiac operations: A randomized prospective trial[J]. The Journal of Thoracic and Cardiovascular Surgery 111: 637-641.
- Mason-DS, Sapala JA, Wood MH, Sapala MA (1998) Influence of a Forced Air Warming System on Morbidly Obese Patients Undergoing Roux-en-Y Gastric Bypass. Obes Surg 8: 453-460.
- Lindwall R, Svensson H, Söderström S, Blomqvist H (2010) Forced air warming and intraoperative hypothermia. Eur J Surg 164: 13-16.
- Winkler M, Akça O, Birkenberg B, Hetz H, Scheck T, et al. (2000) Aggressive Warming Reduces Blood Loss During Hip Arthroplasty. Anesth Analg 91: 978-984.
- Wei YR (2012) Bair hugger Temperature control instrument in the study on the role of surgery in neonates. National Medical Frontiers of China 7: 81-82.
- Ng V, Lai A, Ho V (2006) Comparison of forced-air warming and electric heating pad for maintenance of body temperature during total knee replacement. Anaesthesia 61: 1100-1104.
- Butwick AJ, Lipman SS, Carvalho B (2007) Intraoperative Forced Air-Warming During Cesarean Delivery Under Spinal Anesthesia Does Not Prevent Maternal Hypothermia. Anesth Analg 105: 1413-1419.
- Leung KK, Lai A, Wu A (2007) A randomised controlled trial of the electric heating pad vs forced-air warming for preventing hypothermia during laparotomy. Anaesthesia 62: 605-608.
- De Witte JL, Demeyer C, Vandemaele E (2010) Resistive-Heating or Forced-Air Warming for the Prevention of Redistribution Hypothermia. Anesthesia & Analgesia 110: 829-833.
- Chung SH, Lee BS, Yang HJ, Kweon KS, Kim HH, et al. (2012) Effect of preoperative warming during cesarean section under spinal anesthesia. Korean journal of Anesthesiol 62: 454-460.
- Song RY (2016) Effect of prewarming combined with intraoperative active warming on early postoperative cognitive function recovery in elderly patients with thoracic surgery. HeBei North University, China.
- Wang YJ (2016) The effect of forced-air warming blanket on the patients undergoing total hysterectomy during operative procedures, Suzhou university, China.
- Liu C (2018) Clinical study of the effect of different body temperature on postoperative cognitive function in elderly patients. Xinan medical university, China.
- Liang H (2016) Effectiveness of forced-air warming system in preventing hypothermia and reducing intraoperative blood loss for patients undergoing hip replacement surgery. HeBei North University, China.
- Horn EP, Schroeder F, Gottschalk A (2002) Active Warming During Cesarean Delivery. Anesth Analg 94: 409-414.
- Shin KM, Ahn JH, Kim IS, Lee JY, Kang SS (2015) The efficacy of pre-warming on reducing intraprocedural hypothermia in endovascular coiling of cerebral aneurysms. BMC Anesthesiol 15: 8.
- Qu DC (2016) Effects of air warming blanket on the recovery period of anesthesia in elderly patients undergoing total knee replacement. Dalian medical university, China.
- Ng SF, Oo CS, Loh KH, Lim PY, Chan YH, et al. (2003) A Comparative Study of Three Warming Interventions to Determine the Most Effective in Maintaining Perioperative Normothermia. Anesth Analg 96: 171-176.
- Lai ZY, Li GH, Li K, et al. (2017) Effect of perioperative insulation on patients undergoing total knee arthroplasty. Jiangxi Medical Journal 3: 231-233.
- Murat I, Bernière J, Constant I (1994) Evaluation of the efficacy of a forced-air warmer (bair hugger) during spinal surgery in children. J Clin Anesth 6: 425-429.
- Pei L, Huang Y, Xu Y, Sang X, et al. (2018) Effects of Ambient Temperature and Forced-air Warming on Intraoperative Core Temperature. Anesthesiology 128: 903-911.
- Onik GM, Chambers N, Chernus SA, Zemel R, Atkinson D, et al. (1993) Hepatic cryosurgery with and without the Bair Hugger. J Surg Oncol 52: 185-187.
- Yuan GJ (2013) Study on the effect of different thermal insulation measures on the change of body temperature in patients undergoing laparotomy. Tianjin Medical University, China.
- Hooven K (2011) Preprocedure Warming Maintains Normothermia Throughout the Perioperative Period: A Quality Improvement Project. J Perianesth Nurs 26: 9-14.
- Xu H, Wang Z, Guan X, Lu Y, Malone DC, et al. (2020) Safety of intraoperative hypothermia for patients: meta-analyses of randomized controlled trials and observational studies. BMC Anesthesiol -20: 202.
Citation: Xu H, Lu Y, Guan X, Ma Y, Ma A, et al. (2021) Efficacy and Safety of Forced-Air Warming System versus Passive Warming Measures in Major Surgeries: A Systematic Review. J Anesth Clin Care 8: 65.
Copyright: © 2021 He Xu, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

