
Synthesis and characterization of Al2O3 nanoparticles from Trachyspermum ammi
*Corresponding Author(s):
Eshrat BibiDepartment Of Chemistry, Faculty Of Science, Lahore Garrison University, Pakistan
Email:eshratbibi80@gmail.com
Abstract
Green synthesis approaches are acquiring importance due to their environmentally safe, commercial scalability, versatility, cost-effectiveness, simplicity and efficiency on large scale synthesis. The extract of Trachyspermum ammi was used for the synthesis of Al2O3 nanoparticles. The green synthesized Al2O3 nanoparticles were characterized by using by UV-Vis, FTIR, SEM, XRD and EDX. The green synthesized Al2O3 nanoparticles were characterized by using by UV-Vis, FTIR, SEM, XRD and EDX. The absorbance at 345.4nm from UV-visible spectroscopy shows the formation of Al2O3 nanoparticles. The FTIR results indicated the presence of functional groups responsible for the formation of nanoparticles and the peak between 710.71cm-1 and 871.92 cm-1 confirm the presence of Aluminium oxide. XRD analysis revealed the formation of Al2O3 nanoparticles with average particle size 25.7nm calculated from Scherer formula. EDX analysis demonstrated the elemental composition. The phytochemical screening of Trachyspermum ammi was performed. The antibacterial activity of prepared Al2O3 nanoparticles was investigated by Well diffusion method. Plant extract consist of numerous biochemical and phytochemical compounds that serve as capping and reducing agent that facilitates the production of non-toxic nanoparticles that are useful for pharmaceutical and biological applications.
Keywords
Aluminium hydroxide, Al2O3 nanoparticles synthesis, Trachyspermum ammi
Introduction
Energy and environment are the two main components due to which life exists on the earth and their connection with technology revolutionized the life in the advanced era. Similarly, the nano technology is associated with the nano materials and chemicals (metals and non- metals) present in the environment [1]. The new properties are manipulated in the complexes that are totally changed from basic atoms or molecules. The altered properties shows its functionality, optical properties, their dimensional properties, magnetic properties and even the chemical properties. The nano technology plays a vital role in economic growth and sustainable growth of life [2]. In general, the characteristics of nano-objects are influenced by their chemical composition, as well as their size, shape, composition, and surroundings, as well as their three-dimensional dispersal. It is obvious that production processes have a significant impact on the characteristics of nano-objects. Synthesis methods can be used in a variety of ways.
There are two types of strategies: bottom-up and top-down [3]. Many disadvantages, including as the existence of poisonous organic solvents, the generation of dangerous side-products and intermediate chemicals, and the large energy consumed, make chemical and physical NP synthesis difficult to scale up to large-scale manufacturing. Metal nanoparticles are commonly synthesized in aqueous environments, however due to the existence of attractive forces, such as van der Waal’s, those particles may agglomerate. To avoid particle aggregation and regulate nanoparticle shape, several synthetic compounds known as "capping agents" were added to the steric stabilizers. Other issues with NP production include toxicity, decreased particle synthesis rate, structural particle deformation, and particle growth inhibition. Furthermore, chemical NP production in nanocomposites or metallic NPs is made up of several chemical sorts (atoms) or fragments, which might enhance constituent’s reactivity, as well as affect human healthiness and the atmosphere. On the basis of the large number of molecules and atoms amalgamations used during Nano particles chemical production, it is prerequisite to consider all the information of the reaction conditions. This fact should be a prerequisite to track the molecular species involved along the kinetics of reactions [4]. Biochemical NP production, on the other hand, is easy, cost-effective, more repeatable, and has well-defined physicochemical characteristics, in addition to being ecologically benign. Organic, Inorganic, and hybrid bio NPs are of the three types. Maximum macro-molecules and the cellular structures can generate NPs in response to environmental circumstances and sample treatment [5]. Oxides and metallic bio NPs are two types of inorganic bio NPs. The fabrication of Inorganic Bio NPs is generated by undefined reducing agents in the media by activating the system into the cell to decrease deadliness. The benefits of using biological patterns is the large variety of tridimensional (3D) bio configurations that can be used as originals to generate NPs with a wide range of characteristics and properties [5]. Eco-friendly chemistry also called as green chemistry that deals with the alteration of biological complexes to green chemicals which is further modified by the nanomaterial particles without using the toxicity of chemicals that is biosynthesized chemicals [6].
Metal oxide when manufactured from plants i.e. synthesized through green synthesis, the phytochemicals present in plants act as reducing agent while the metal Nano particles act as stabilizer [7]. The production of Aluminum oxide nanoparticles can be done in a variety of ways (Al2O3 NPs or Nano alumina). The solution combustion technique is an energy-efficient and environmentally benign approach for producing low-cost nano adsorbents [8]. Trachyspermum ammi (Umbelliferae), also known as Ajwain in India, is found across the country's northwestern regions [9]. Trachyspermum ammi is a well-known source of components with promise bioactivity that can be used in pharmaceuticals. T. ammi is an yearly grown herb that grows up to the 90 cm tall and is endemic to Egypt's desert and less desert areas [10]. They have stimulating, antispasmodic, and the carminative qualities and have been utilized in the cure of pomposity, atonic indigestion, abdominal tumors, diarrhea, stomach aches, piles, and bronchial difficulties, lack of appetite, asthma, and amenorrhea for centuries. The fruits consists 2–5% Ajwain oil, a brown-colored essential oil that is accountable for the plant's odor and flavor. It's used to cure gastric and intestinal disorders, a lack of craving, and respiratory issues [11].
Materials And Methods
Optimized concentration of spice T. Ammi about 100g dissolved in 1000ml distilled water and continue to heating on burner for 1hour, until the 2/3rd volume of extract was left behind from total amount added distilled water. Extract was cooled and filtered using filter paper (Whattman No. 1). Now, extract is ready for use. The extract was freshly prepared for the synthesis of Al2O3 NPs. Al2O3 nanoparticles was synthesized by Co-precipitation method. In first step, Sodium hydroxide was mixed with extract and Aluminium chloride react with Sodium hydroxide. In second step, Aluminium hydroxide leads to formation of Al2O3 nanoparticles. In this method extract acts as reducing agent.
Procedure
For the preparation of Al2O3NPs, 60g of AlCl3 was dissolved in 200ml distilled water and 180g of NaOH in 200ml distilled water in ratio of 1:3. The extract was mixed with AlCl3 solution in the beaker at 25°C and was put in funnel. On lower side, NaOH in a flask on hotplate, funnel on upper side and thermometer hang in it. The extract and AlCl3 solution fell drop by drop from funnel into NaOH solution which is on hotplate. Both the solutions of AlCl3 and NaOH were mixed with magnetic stirrer. The temperature which was recorded at this time between 60-80°C. At this point, the formation of nanoparticles was started. The second step of synthesis is washing. After centrifugation, the mixture was left for 24 hours. Next day, washing was done. During washing small quantity of ethanol was also added for the removal of some extra impurities. Last step is the filtration and drying. Dried NPs were obtained after 72 hours at room temperature, followed by calcined at 450°C for three hours in muffle furnace and then subjected for the characterization of Al2O3NPs.
Results And Discussion
UV- Visible Spectroscopy: UV-Visible spectroscopy is an important technique used for the characterization of nano particles (Figure 1). Its principal is chased an absorption of UV-Visible radiation. The absorbance peak from 200nm to 400nm show presence of nano particles. The broad band is observed of Aluminum Oxide towards blue shift at 345.4nm [12]. A smaller absorption edge is advantageous for simpler electronic conductivity (greater energy transformations) and consequently quicker ionic conductivity.
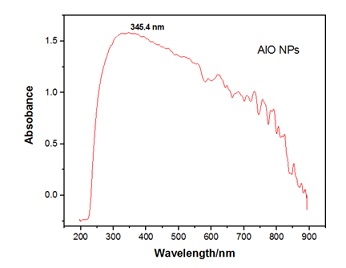 Figure 1: UV/Visible spectrum
Figure 1: UV/Visible spectrum
FTIR Analysis: FTIR is a time-saving, non - destructive approach for detecting a variety of functional groups and is responsive to fluctuations in chemical composition. The information provided by FTIR is influenced by the chemical composition and physical condition of the entire sample [13]. Figure 2 given below shows the spectrum of FTIR of biosynthesized Al2O3 Nanoparticles using Trachyspermum ammi. The absorption bands are observed. The bands are at 710.71cm-1, 871.08cm-1, 1406.23cm-1 and 3366.11cm-1. The peak present at 710.71cm-1 shows the C=C bending of 1,2-disubstituted diene (cis) while 3366.11cm-1 shows the N-H stretching of Aliphatic primary amines (Table 1). The peat at 1406.23cm-1 shows the O-H bending of alcohols. The broad curve between 710.71cm-1, 871.08cm-1 and 1021.12cm- 1 shows the bending of Aluminum oxide. The peak at 1021.12cm-1 Al bending of biosynthesized Nano particles [14].
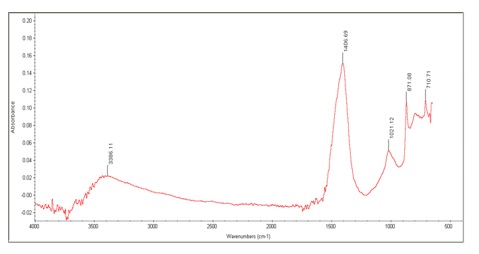 Figure 2: FTIR Spectrum
Figure 2: FTIR Spectrum
|
IR Ranges (cm-1) |
Functional groups |
Compound class |
|
710.71 |
C=C bending |
1,2 disubstituted dienes (cis) |
|
871.08 |
Al bending |
Al2O3 NPs |
|
1406.23 |
H bending |
Alcohol |
|
3366.11 |
N-H Stretching |
Amine compounds |
Table 1: Evaluation of different functional groups in FTIR spectrum
Scanning Electron Microscope (SEM): Scanning electron microscope is used for investigating topographies of nano particles at very high magnifications. It's a sort of electron microscope that scans the surface with a focused stream of electrons to obtain pictures of nano particles. A grid scan pattern is used to scan the electron beam, and the location of the beam is coupled with the intensity of the received signal to create a picture. The patterns of Aluminum oxide nano structures were depicted in the picture of SEM examination (Figure 3).
 Figure 3: SEM Analysis of Al2O3
Figure 3: SEM Analysis of Al2O3
When subjected to the energetic electrons of the SEM, the bright regions of the picture demonstrate a strong generation of secondary electrons. Because of the high specific surface area ratio in certain places, this is the case. SEM images showed that Aluminum oxide Nano-particles are almost spherical in shape (Figure 4), irregular and also showed agglomeration of disc shaped nanoparticles.
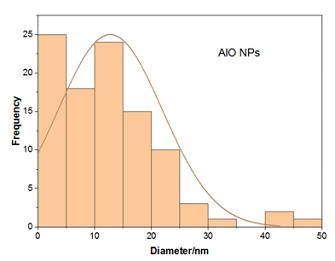 Figure 4: Size distribution ratio of Al2O3 NPS in nm.
Figure 4: Size distribution ratio of Al2O3 NPS in nm.
X-Ray Diffraction (XRD): XRD is a technique for determining the lattice parameters of a substance. XRD pattern of synthesized Al2O3 nanostructures using the extract of Trachyspermum ammi is shown in Figure 5. The intensive peak were observed at 2θ = 26.5°c with (002) orientation. While rest of peak at 2θ= 29.4, 38.96and 47.92 are corresponds to the planes (101), (102) and (220). (Table 2) These mentioned peaks are in agreement with the standard JCPDS card number then the nan particle size was determined by using the data from the XRD pattern by using the Scherer formula
D=kλ/βcosθ
Where, D = crystallite size
λ = wavelength of the X-rays produced in the machine β = width of a peak at half of its intensity
θ = angle of the corresponding peak k = shape factor
The average crystallite size was found to be about 25.7nm
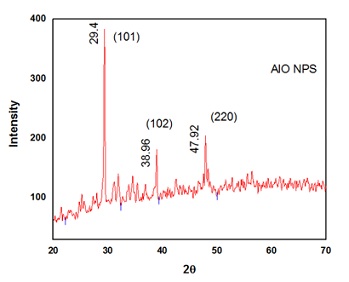 Figure 5: XRD Analysis ofAl2O3
Figure 5: XRD Analysis ofAl2O3
|
K |
Λ |
Peak Position 2θ |
β (FWHM) |
crystallite size (L) |
|
|
(Å) |
Θ |
Θ |
nm |
|
0.94 |
1.54178 |
29.4 |
0.25575 |
33.56685 |
|
0.94 |
1.54178 |
38.96 |
0.38196 |
23.05976 |
|
0.94 |
1.54178 |
47.92 |
0.42713 |
21.27393 |
Table 2: Particle size of Al2O3 nano particles from XRD data
Energy-Dispersive X-ray Spectroscopy (EDX/EDS): Energy dispersive X-Ray spectroscopy is used to analyze the structure and analytical constitution of substances [15]. EDX spectroscopy is used to determine the chemical compositions of a material and use a SEM. EDX can identify elements qualitatively and quantitatively [16]. EDX spectrum shows the presence of Al and O in the biosynthesized Aluminum oxide nano particles as shown in the Figure 6. The peaks other than Al and O showed the presence of impurity (Table 3) [17].
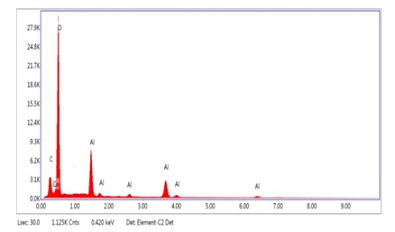 Figure 6: EDS Spot 1
Figure 6: EDS Spot 1
|
Element |
Weight % |
Atomic % |
|
C |
11.50 |
17.52 |
|
O |
50.09 |
57.32 |
|
Al |
30.22 |
21.318 |
|
Ca |
8.19 |
3.842 |
|
Total |
100.0 |
100.0 |
Table 3: Elemental ratio of Al2O3 Nano particles
Biological Potential of Al2O3 nanoparticles: Antibacterial activity of Al2O3 nanoparticles was done against two bacterial strains (E.coli, S. aureus). Antibacterial potential of plant extract were determined by using well diffusion method.
Well Diffusion Assay: By using a well diffusion experiment against (E.Coli, S. Aureus), the antibacterial activity of Al2O3 nanoparticles were characterized with little amendments (Table 4). Powdered Al2O3 nanoparticles were diluted 1 percent DMSO to make Al2O3 nanoparticles solution for antibacterial activity. Similarly, solution of antibiotic was prepared by adding 100mg of antibiotic (Oxytetracycline) in one mg of distilled water. We used the backside blue micropipette tips to make two wells in the agar plate after inoculation. Aluminium oxide nanoparticles (100L) were then added to one well and antibiotic to another well. The plates were then incubated at 370C for 18 to 24 hours. The zone of inhibition around the wells was seen after incubation, and the zone diameter was measured in millimeters (mm) with the ruler. It has previously been observed that as the concentration of nanoparticles increases, so does the inhibition of growth of bacteria takes place [18].
|
Bacterial strain |
DIZ (mm) |
|
|
Al2O3 NPs. |
Oxytetracycline |
|
|
E.Coli |
18±2.06 |
19±2.06 |
|
S. aureus |
16±1.86 |
17±1.86 |
Table 4: Antibacterial activity of Al2O3 nanoparticles
The zones of inhibition are given in the table. The zone of inhibition for E.coli and S. aureus is at 18 and 16 respectively. The highest diameter of inhibition zone of Al2O3 nanoparticles was found to be 18mm against E. coli.
Conclusions
Trachyspermum ammi was used to successfully manufacture and analyze aluminium oxide nanoparticles. These nanoparticles' functional groups, crystalline structure, crystalline size, morphologies, and characteristics were compared to those of ordinary aluminium oxide nanoparticles. Then these nanoparticles are characterized by different techniques UV-Vis, FTIR, XRD, EDX and SEM. UV-visible spectroscopy shows the maximum absorption at the wavelength of 345.5nm. FTIR shows the Al2O3 nanoparticles peaks between 871.08cm-1 and 1021.12cm-1. SEM and XRD analysis shows that they are spherical and oval shape having crystallite size of 25.7nm. EDX shows the elemental analysis and the impurity that is Calcium. Phytochemicals analysis done by using different chemical reagents that shows the presence of phyto-constituents in Trachyspermum ammi. Antibacterial potent of different bacterial strains were determined by using well diffusion assay. The natural mechanism would be more exquisite in manufacturing a range of other significant ceramic nanoparticles if it remained connected to green chemistry principles, practical efficiency, and the mildness of the reaction, economic viability, and biomedical application. In future we can make perfect material for Far-Infra red material, improve the plastic wear industry, decreasing the density of ceramic materials and in aerospace materials for making aircraft wings leading edges.
References
- Sun H (2019) Grand challenges in environmental Frontiers in Nanotechnology 1: 2.
- Basiuk VA, Basiuk EV (2015) Green processes for nanotechnology. Springer
- Schwarz JA, Contescu CI, Putyera K (2004) Dekker encyclopedia of nanoscience and nanotechnology
- Tolaymat TM, Badawy EAM, Genaidy A, Scheckel KG, Luxton TP, et al. (2010) An evidence-based environmental perspective of manufactured silver nanoparticle in syntheses and applications: a systematic review and critical appraisal of peer-reviewed scientific Science of the total environment 408: 999-1006.
- Durán N, Marcato PD (2011) Biotechnological routes to metallic nanoparticles production: mechanistic aspects, antimicrobial activity, toxicity and industrial applications Nano-antimicrobials : 337-374.
- Nasrollahzadeh M, Sajjadi M, Sajadi SM, Issaabadi Z (2019) Green nanotechnology. Interface science and technology 28: 145-198.
- Ghotekar S (2019) Plant extract mediated biosynthesis of Al2O3 nanoparticles-a review on plant parts involved, characterization and Nanochemistry Research 4: 163-169.
- Prabhakar R, Samadder SR (2018) Low cost and easy synthesis of aluminium oxide nanoparticles for arsenite removal from groundwater: a complete batch study. Journal of Molecular Liquids 250: 192-201.
- Ramaswamy S, Sengottuvelu S, Sherief SH, Jaikumar S, Saravanan R, et al. (2010) TRACHYSPERMUM AMMI International Journal of Pharma and Bio Sciences 1: 1.
- Ashraf M (2002) Salt tolerance of cotton: some new advances. Critical Reviews in Plant Sciences 21: 1-30.
- Bairwa R, Sodha RS, Rajawat BS (2012) Trachyspermum ammi. Pharmacognosy reviews 6: 56-60.
- Nduni MN, Osano AM, Chaka B (2021) Synthesis and characterization of aluminium oxide nanoparticles from waste aluminium foil and potential application in aluminium-ion cell. Cleaner Engineering and Technology 3: 100108.
- Cocchi M, Foca G, Lucisano M, Marchetti A, Pagani MA, et al. (2004) Classification of cereal flours by chemometric analysis of MIR Journal of agricultural and food chemistry 52: 1062-1067.
- Chu TPM, Nguyen NT, Vu TL, Dao TH, Dinh LC, et al. (2019) Synthesis, characterization, and modification of alumina nanoparticles for cationic dye removal. Materials 12:
- Zadora G, Brozek MZ (2003) SEM–EDX—a useful tool for forensic Materials chemistry and physics 81: 345-348.
- Mutalib MA, Rahman MA, Othman MHD, Ismail AF, Jaafar J (2017) Scanning electron microscopy (SEM) and energy-dispersive X-ray (EDX) spectroscopy Membrane characterization : 161-179.
- Sumesh KR, Kanthavel K (2019) Green synthesis of aluminium oxide nanoparticles and its applications in mechanical and thermal stability of hybrid natural composites. Journal of Polymers and the Environment 27: 2189-2200.
- Fakeh MSA, Alsaedi RO (2021) Synthesis, Characterization, and Antimicrobial Activity of CoO Nanoparticles from a Co (II) Complex Derived from Polyvinyl Alcohol and Aminobenzoic Acid The Scientific World Journal, Egypt.
Citation: Bibi E (2023) Synthesis and characterization of Al2O3 nanoparticles from Trachyspermum ammi. J Agron Agri Sci 6: 050
Copyright: © 2023 Eshrat Bibi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

