
The Impact of Early Pulmonary Rehabilitation on the Multidimensional Aspects of Dyspnea and Exercise Performance Following Acute Exacerbation of Chronic Obstructive Pulmonary Disease: A Randomized Trial
*Corresponding Author(s):
Amr Mounir ShoukriDepartment Of Chest Diseases, Faculty Of Medicine, Ain Shams University, Egypt
Tel:+201006601870,
Email:amr_shoukri@hotmail.com
Abstract
Background: Acute Exacerbations of Chronic Obstructive Pulmonary Disease (AECOPD) are associated with severe dyspnea and exercise intolerance. Early Pulmonary Rehabilitation (EPR) may lead to improvements in dyspnea and exercise tolerance, as it does in stable COPD patients.
Objective: To investigate the potential benefits of EPR, following AECOPD, in terms of multidimensional aspects of dyspnea and exercise performance.
Methods: One hundred and six patients admitted in a university hospital with AECOPD were randomized after discharge to either EPR for 8 weeks (EPR group) or Usual Care (UC) (UC group). All patients carried out the following tests, initially and after 8 weeks: spirometry, 6 Minute Walk Test (6MWT), and a symptom-limited incremental cycle Cardiopulmonary Exercise Test (CPET), and different dyspnea dimensions evaluation as following: Dyspnea intensity during incremental exercise using Borg scale, dyspnea 12 questionnaire and COPD Assessment Test (CAT) to assess sensory perceptual, affective distress, and symptom impact domains respectively.
Results: Forty male patients in each group well matched for age, body mass index, smoking index and spirometry completed the study. Significant improvements were detected following EPR in different dyspnea domains (sensory-perceptual, affective, and impact domains), and exercise performance and endurance. A highly significant difference (P=0.0001) was found in the magnitude of improvement of 6MWT (32.75 meters), CAT score (0.37), dyspnea 12 questionnaire (0.68), Borg scale during incremental exercise (0.31), and CPET duration (15.86 seconds), in the EPR group compared to UC group.
Conclusion: EPR following AECOPD was associated with clinically significant improvements in different domains of dyspnea, and exercise performance and endurance.
Keywords
TRIAL REGISTRATION
Trial Registration: ClinicalTrials.gov NCT03611127.
Date of registration: August 2, 2018.
Retrospectively registered
BACKGROUND
Acute Exacerbations of Chronic Obstructive Pulmonary Disease (AECOPD) are associated with severe dyspnea, activity restriction, accelerated physiological impairment and increased mortality [1]. These events are usually associated with worsening expiratory flow limitation. These, in combination, give rise to acute Dynamic Hyperinflation (DH) and dyspnea [1]. Respiratory muscle function is negatively affected by DH, functional weakening of the diaphragm is also encountered, and development of respiratory acidosis occurs, requiring hospital admission [1].
Following hospital discharge for an acute exacerbation, patients are typically more breathless and less active, and they may remain so for many weeks [2].
Interventions designed to fasten the recovery and improve COPD patients’ symptoms after hospital admission have many benefits, it lead to significant improvement in functional performance, and hence the quality of life of those patients, and it may also lead to reduction in further hospital admissions and future health care utilization [3].
Pulmonary Rehabilitation (PR), an intervention based on individually tailored exercise training, has emerged as arguably the most effective non-pharmacological intervention in improving dyspnea, exercise capacity and health status in COPD patients [4,5].
An Early Pulmonary Rehabilitation (EPR) following hospital discharge after AECOPD may have several benefits, exercise training is expected to decrease the ventilatory requirements during physical effort, which will reduce DH that leads to exercise limitation [6-9]; this, in turn, would be expected to be associated with an improvement of the three major dimensions of dyspnea: the sensory-perceptual domain, the affective distress, and the symptom impact or burden.
Thus, we aimed to determine the potential physiological and clinical impact of EPR on the multidimensional aspects of dyspnea (sensory perceptual, affective distress, and symptom impact domains) and exercise performance in COPD patients following hospital discharge from AECOPD.
SUBJECTS AND METHODS
Study Subjects: Hospitalized COPD patients with a diagnosis of AECOPD with no clinically significant arterial hypoxemia at rest or on exercise (resting percutaneous oxygen saturation (SpO2)>90% or a sustained decrease of <4% during exercise) were recruited. Diagnosis of COPD, AECOPD and spirometric assessment of airflow limitation severity was based on Global Initiative for Chronic Obstructive Lung Disease (GOLD) [10]. Patients with a prior diagnosis of other cardiorespiratory conditions (i.e., bronchial asthma, interstitial lung diseases, primary pulmonary hypertension, chronic congestive heart failure), as well as other conditions such as orthopedic, muscular and peripheral vascular diseases that could cause or contribute to breathlessness and exercise intolerance and/or could interfere with carrying out of exercise testing, were excluded.
Ethics, Consent and Permissions:
- Ethics approval from Ain Shams University, Faculty of Medicine research ethics committee (FWA00017585).
- Written informed consent signed by each patient to participate in the study.
- Consent obtained from each participant to publish his data collected in the study.
Study Design: The study was conducted in the Chest department of Ain Shams University hospital, Cairo, Egypt. After receiving ethical approval from Ain Shams University, Faculty of Medicine research ethics committee (FWA00017585), informed consent obtained from all participants. Each participant performed 2 visits. At visit 1 (within one week following hospital discharge), patients were familiarized with dyspnea and quality of life questionnaires/scales and carried out spirometric lung function test, 6MWT, and a symptom-limited incremental cycle Cardiopulmonary Exercise Test (CPET). Then, simple randomization method using coin flipping was done by the principal investigator, patients were randomized to either EPR or Usual Care (UC), both were receiving their standard maintenance therapy, the patients and investigators were not blinded to allocation due to the nature of the intervention and the participation of investigators in the pulmonary rehabilitation process. Eight weeks rehabilitation program offered to EPR group only, featuring two directly supervised sessions per week, each lasting 2 hours, in accordance with recommended and well described exercise-training programs [4,11]. At visit 2, conducted 8 weeks after visit 1, all patients completed spirometric lung function test followed by an incremental CPET, dyspnea and quality of life questionnaires/scales. Subjects continued their respiratory medications except for short-acting bronchodilators prior to exercise testing (short-acting B2-agonists=4 hours, short-acting anticholinergics=6 hours). Subjects were asked to avoid smoking for at least 60 minutes prior to each visit, caffeine-containing beverages and heavy meals at least 4-6 hours prior to testing, as well as strenuous physical exertion for at least 12 hours before each visit day. Visits were conducted at the same time of the day for each subject. The study adheres to CONSORT guidelines and include a completed CONSORT checklist as an additional file.
Methods:
Spirometric lung function test: Assessment of baseline spirometry according to recommended techniques [12,13].
Dyspnea evaluation: Dyspnea was assessed before and after (visit 1 and 2) EPR program by evaluating its multidimensional aspects which comprise three major dimensions: the sensory-perceptual domain, the affective distress domain, and the symptom impact/burden domain [12,13].
The sensory-perceptual dimension, which includes ratings of dyspnea intensity and its quality (that is, “how breathing feels like”), was evaluated by using the Borg scale during incremental cycle exercise [14]. The affective distress domain, which addresses the question of “how distressing breathing is” and focuses on the perception of immediate unpleasantness or the cognitive evaluative response about the potential consequence of what is perceived, was assessed by using the dyspnea-12 questionnaire [15]. The symptom impact/burden dimension, which evaluates how dyspnea impacts on functional ability/disability, health status, and quality of life, was evaluated by using the COPD Assessment Test (CAT) [16].
Exercise Testing: Cardiopulmonary Exercise Tests (CPETs) were conducted on an electronically braked cycle ergometer in accordance with recommended techniques and previously published studies. All incremental exercise tests consisted of a steady-state resting period of 6 minutes and a 3-min warm-up of unloaded pedaling followed by an incremental test in which the work rate (WR) increased at 1-minute intervals (ramp protocol) by increments of 5 watts until the point of symptom-limitation (peak exercise) [8,12,13,17,18]. Patients were instructed to maintain the pedaling rate between 50 and 70 revolutions per minute. Breath-by-breath data were collected at baseline and throughout exercise while subjects breathe through a mouthpiece with attached low-resistance flow transducer with nasal passages occluded by a nose-clip: Minute volume (VE), oxygen uptake (VO2), carbon dioxide production (VCO2), end-tidal carbon dioxide partial pressure (PETCO2), tidal volume (VT), respiratory frequency (Rf) were calculated. Electrocardiographic monitoring of heart rate (HR), rhythm, ST-segment changes, blood pressure by indirect sphygmomanometry, and percutaneous oxygen saturation (SpO2) by pulse oximetry were carried out continuously throughout exercise testing [8,12,13,17,18]. During incremental CPET, the Ventilatory Anaerobic Threshold (VAT), and VE/VCO2 slope were calculated in accordance with recommended techniques and previously published studies [12,17].
Early pulmonary rehabilitation: The pulmonary rehabilitation program consisted of two supervised sessions per week for eight weeks. Each session lasted two hours: One hour of exercise training, conducted in 4 sets of aerobic walking and cycling, the initial exercise level was set at a work rate corresponding 70% of peak VO2 from the baseline CPET, and progressively increased by 5 Watt, until 80% of the baseline peak VO2 is reached, each set is 10 minutes. Strength training for the upper and lower limb was also applied. The second hour was dedicated to education concerning the disease (COPD) and its management, smoking cessation, nutrition and other lifestyle issues. Patients were also encouraged to perform daily home exercise of at least 20 minutes of ground walking. Any encountered side effects or complications related to EPR were recorded.
Blinding: The patients and investigators were not blinded to allocation due to the nature of the intervention and the participation of investigators in the pulmonary rehabilitation process.
Sample size calculation: The primary outcome measure of the study was to determine the effect of early pulmonary rehabilitation on the different aspects of dyspnea, as well as exercise performance in COPD patients following exacerbation. Sample size was calculated using STATA program, setting the type-1 error (α) at 0.05 and the power (1-β) at 0.8. On the basis of results from a previous study [3], calculation of sample size estimated that33 cases per group is needed, and with taking in consideration 20% drop out rate, the needed sample is 40 cases per group.
Statistical analysis: The collected data was revised, coded, tabulated and introduced to a PC using Statistical package for Social Science (IBM Corp. Released 2011. IBM SPSS Statistics for Windows, Version 20.0. Armonk, NY: IBM Corp). Quantitative non-parametric variables are expressed as mean and SD, Median and Interquartile Range (IQR). Qualitative variables are expressed as frequencies and percent. Student t test and Mann Whitney Test were used to compare a continuous variable between two study groups. Chi square test and Fisher’s exact test were used to examine the relationship between Categorical variables. Paired t test was used to assess the statistical significance of the difference between two means measured twice for the same study group. A P-value< 0.05 was considered statistically significant.
RESULTS
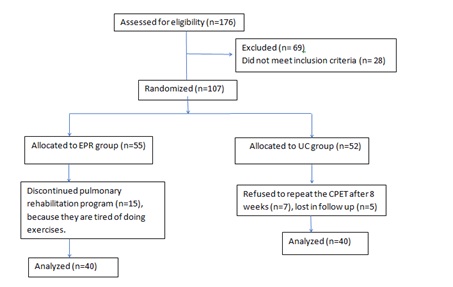
All the participants were males, and both groups were well matched for age, body-mass index, and smoking status (Table 1 and Figure 1).
|
Socio-demographic data |
Group |
|||
|
EPR |
UC |
|||
|
Mean |
±SD |
Mean |
±SD |
|
|
Age |
57.4 |
7.93 |
58.43 |
7.72 |
|
Weight (kg) |
74.33 |
13.36 |
72.58 |
8.8 |
|
Height (cm) |
170.98 |
7.24 |
172.73 |
7.96 |
|
25.36 |
4.25 |
24.33 |
3.08 |
|
|
28.2 |
9.96 |
30.32 |
9.99 |
|
|
Sex : Male (n %) |
40 |
100.00% |
40 |
100.00% |
|
COPD spirometric GOLD classification |
|
|
|
|
|
GOLD 3 [30%≤FEV1<50%predicted]# (n %) |
11 |
27.50% |
15 |
37.50% |
|
GOLD 4 [FEV1<30% predicted]# (n %) |
29 |
72.50% |
25 |
62.50% |
|
Clinical assessment parameter |
|
|
|
|
|
CAT Score |
12.55 |
3.09 |
11.35 |
2.76 |
|
6-minute walk test (METERS) |
336.25 |
69.97 |
360 |
84.79 |
|
Dyspnea 12 questionnaire |
17.05 |
5.49 |
16.15 |
6.6 |
|
Pulmonary function test |
|
|
|
|
|
FEV1 (L) |
1.88 |
0.5 |
1.83 |
0.59 |
|
FEV1 % PREDICTED |
57.45 |
15.5 |
53.49 |
15.43 |
|
FVC (L) |
3.35 |
0.61 |
3.27 |
0.62 |
|
FVC % PREDICTED |
74.2 |
13.81 |
73.26 |
12.07 |
|
FEV1/FVC % |
55.95 |
8.03 |
54.68 |
9.34 |
|
MEF 50 (%) |
34.03 |
13.5 |
32.23 |
12.88 |
|
VE max (L/min) |
43.1 |
12.31 |
46.72 |
13.59 |
|
BMI: Body mass index |
||||
No significant difference between the two studied groups as regard the number of patients at each COPD spirometric GOLD stage. In EPR group, 11 patients were in GOLD stage 3 and 29 in GOLD stage 4, and in UC group, 15 patients were in GOLD stage 3 and 25 in GOLD stage 4 (Table 1).
No significant difference detected between the EPR and UC group in the results of initial assessment which included COPD assessment test (CAT), dyspnea 12 questionnaire, 6MWT , spirometric lung function test (Table 1), and cardiopulmonary exercise tests (Table 2). The CAT score was 12.55±3.09 for EPR group and 11.35±2.76 for UC group, dyspnea-12 questionnaire was 17.05±5.49 for EPR group and 16.15±6.60 for UC group. Six-minute walk test results were 336.25±69.97 for EPR group and 360±84.79 for UC group.
|
Cardiopulmonary exercise test parameter |
Group |
P* |
Sig |
|||
|
EPR |
UC |
|||||
|
Mean |
±SD |
Mean |
±SD |
|||
|
Borg scale during incremental exercise |
7.58 |
0.9 |
7.85 |
0.95 |
0.188 |
NS |
|
VO2 (ml/min/Kg) |
18.68 |
8.72 |
18.58 |
9.17 |
0.962 |
NS |
|
VAT or VO2 at VT (ml/min/Kg) |
16.7 |
8.33 |
16.69 |
8.52 |
0.997 |
NS |
|
Resting PETCO2 (mmHg) |
36.53 |
1.34 |
36 |
1.15 |
0.064 |
NS |
|
Peak RER |
1.05 |
0.09 |
1.03 |
0.1 |
0.434 |
NS |
|
Resting SpO2 (%) |
98.07 |
0.92 |
98.37 |
0.7 |
0.105 |
NS |
|
Minimum SpO2 (%) |
95.48 |
1.04 |
95.3 |
1.11 |
0.469 |
NS |
|
Resting RR (breath/minute) |
16.93 |
1.7 |
16.3 |
1.54 |
0.089 |
NS |
|
Maximum RR (breath/minute) |
40.13 |
5.73 |
39.8 |
5.72 |
0.8 |
NS |
|
Resting HR (beat/minute) |
79.58 |
5.59 |
77.82 |
3.13 |
0.089 |
NS |
|
Peak HR (beat/minute) |
142.68 |
9.67 |
141.25 |
7.96 |
0.474 |
NS |
|
% of age predicted maximal HR |
87.6 |
4.7 |
87.25 |
3.71 |
0.713 |
NS |
|
Maximal work load (Watts) |
120.3 |
14.89 |
115.65 |
11.47 |
0.122 |
NS |
|
Test duration (sec) |
569.73 |
122.53 |
590.75 |
125.44 |
0.451 |
NS |
|
VO2: Oxygen uptake |
||||||
Pulmonary rehabilitation program was performed to EPR group, we monitored good compliance to exercise training, borg scale during the training sets showed a mean of 4.7±0.83.
In each group, we compared the initial and later results. In EPR group, patients showed highly significant clinical improvement (Table 3) after pulmonary rehabilitation. Certain parameters of cardiopulmonary exercise tests also showed highly significant improvement after pulmonary rehabilitation (peak VO2, and test duration), while other parameters were not significantly different (Table 3).
|
Variables |
Mean |
±SD |
P* |
|
Pre CAT Score |
12.55 |
3.08 |
0.002 |
|
Post CAT Score |
11.8 |
2.85 |
|
|
Pre 6 minute walk test (METERS) |
336.25 |
69.97 |
0.0001 |
|
Post 6 minute walk test (METERS) |
368 |
73.2 |
|
|
Pre Dyspnea 12 questionnaire |
17.05 |
5.49 |
0.0001 |
|
Post Dyspnea 12 questionnaire |
15.6 |
4.81 |
|
|
Pre Borg scale during incremental exercise |
7.58 |
0.9 |
0.0001 |
|
Post Borg scale during incremental exercise |
6.55 |
0.9 |
|
|
Pre Peak VO2 (ml/min/Kg) |
18.675 |
8.725 |
0.001 |
|
Post Peak VO2 (ml/min/Kg) |
19.365 |
8.4518 |
|
|
Pre Test duration (sec) |
569.73 |
122.533 |
0.0001 |
|
Post Test duration (sec) |
604.25 |
126.752 |
|
|
CAT : COPD assessment test |
|||
Significant improvement was detected in certain clinical, and cardiopulmonary exercise tests parameters (Table 4), when comparing the results of initial tests and those done after 8 weeks in UC group.
|
Variables |
Mean |
±SD |
P* |
|
Initial CAT Score |
11.35 |
2.76 |
0.006 |
|
CAT Score (after 8 weeks) |
11.52 |
2.87 |
|
|
Initial 6 minute walk test (METERS) |
360 |
84.79 |
0.378 |
|
6 minute walk test (METERS) (after 8 weeks) |
359 |
83.13 |
|
|
Initial Dyspnea 12 questionnaire |
16.15 |
6.6 |
0.006 |
|
Post Dyspnea 12 questionnaire |
16.5 |
6.57 |
|
|
Initial Borg scale during incremental exercise |
7.85 |
0.949 |
0.281 |
|
Borg scale during incremental exercise (after 8 weeks) |
7.73 |
1.012 |
|
|
Initial Peak VO2 (ml/min/Kg) |
18.58 |
9.1653 |
0.038 |
|
Peak VO2 (ml/min/Kg) (after 8 weeks) |
18.173 |
8.6483 |
|
|
Initial Test duration (sec) |
590.75 |
125.441 |
0.098 |
|
Test duration (sec) (after 8 weeks) |
585.75 |
119.462 |
|
|
CAT: COPD assessment test |
|||
We compared both groups regarding the magnitude of change detected in clinical data and cardiopulmonary exercise tests. A highly significant difference in favour of the EPR group regarding the following parameters: CAT score, dyspnea 12 questionnaire, 6-minute walk test, Borg scale during incremental exercise, peak VO2, and CPET duration (Table 5).
|
Variables |
Group |
P* |
|||||||||
|
EPR |
UC |
||||||||||
|
Mean |
±SD |
Median |
IQR** |
Mean |
±SD |
Median |
IQR** |
||||
|
6MWT change |
31.75 |
24.74 |
26 |
0 |
40 |
-1 |
7.09 |
0 |
0 |
0 |
0.0001 |
|
CAT change |
0.75 |
1.43 |
0 |
0 |
1 |
-0.18 |
0.38 |
0 |
0 |
0 |
0.0001 |
|
Dyspnea 12 change |
1.45 |
1.97 |
0 |
0 |
2 |
-0.35 |
0.77 |
0 |
0 |
0 |
0.0001 |
|
Borg scale change |
1.03 |
1 |
1 |
0 |
2 |
0.13 |
0.72 |
0 |
0 |
1 |
0.0001 |
|
VO2 change |
0.69 |
1.25 |
0.4 |
-0.1 |
1.65 |
-0.41 |
1.2 |
0 |
-0.3 |
0 |
0.0001 |
|
VAT change |
-0.02 |
1.15 |
0.05 |
-0.6 |
0.45 |
-0.45 |
1.12 |
-0.15 |
-0.95 |
0.05 |
0.126 |
|
Peak RER change |
0 |
0.08 |
0 |
-0.06 |
0.08 |
0.01 |
0.05 |
0 |
-0.01 |
0 |
0.791 |
|
Work load change |
0.4 |
4.09 |
1 |
-2.5 |
3 |
0 |
0 |
0 |
0 |
0 |
0.301 |
|
Test duration change |
34.53 |
41.06 |
30 |
0 |
70 |
-5 |
18.67 |
0 |
-10 |
0 |
0.0001 |
|
**inter quartile range |
|||||||||||
No side effects or complications related to EPR were recorded in current study.
DISCUSSION
The effect of pulmonary rehabilitation on COPD outcomes was evaluated in multiple evidence-based reviews, and strong evidence has been documented regarding the following aspects: improved exercise performance, dyspnea relief, and improved health related quality of life [19-23]. These beneficial effects were observed even without a direct documented effect on measured lung function, such as forced expiratory volume in one second (FEV1), and this paradox may be explained by the fact that pulmonary rehabilitation treats the systemic effects of COPD and its common comorbidities [4].
The concept of PR as a non-pharmacologic integral component in COPD management is well established and considered as a standard of care for stable COPD patients [24]. However, the effects of EPR in the acute recovery phase after hospital admission for AECOPD needs further validation. Since, the physiological deficits presented clinically by dyspnea and exercise intolerance by patients after a hospital admission with AECOPD are, at least in part, amenable to PR, and their correction may lead to notable improvement in health status, exercise capacity and help in preventing re-exacerbation [3,25].
We noticed that even UC group, who were not subjected to pulmonary rehabilitation, showed significant improvement in two dyspnea dimensions (affective distress and impact domains) when comparing the initial evaluation and that performed 8 weeks later, after an acute exacerbation, and these findings could be explained by the normal recovery process that occurs after the resolution of the acute condition. Other parameters as the sensory perceptual domain of dyspnea, and exercise performance and endurance failed to significantly improve in the UC group. More importantly, comparing the two groups as regard the magnitude of improvement in different dyspnea domains, 6MWT, and exercise performance, we found a significant difference in favor of EPR group,we also detected that these differences were clinically significant, as most of the patients described better performance of their daily activities. indicating the beneficial effect of EPR on dyspnea and exercise performance.
Previous trials that studied the effectiveness of ERP for COPD patients after acute exacerbation showed controversial results [3,25-27].
Certain studies have clearly demonstrated the benefits of EPR, in the recovery period after hospital discharge, on the functional capacity, quality of life and risk of hospital readmission compared to UC [3,25].
Other studies have not found any benefit for implementing EPR for COPD patients after acute exacerbation, they did not detect any enhancement of the recovery of physical function, improvement in the quality of life, or reduction of the risk of re-admission [26,27]. Moreover, one study has reported that mortality at 12 months was higher in the group who underwent EPR, and the authors made their recommendation against the implementation of EPR after AECOPD [26]. These negative results may be explained to some extent by the methodological quality of these studies characterized by the extensiveness of the rehabilitation programs with many training sessions and long duration, in addition to, inclusion of other chronic respiratory diseases (e.g. bronchiectasis, interstitial lung diseases) associated to COPD, in the study with increased morality in EPR group [26].
Many challenges may interfere with implementing EPR in clinical practice. The clinical state of the patient which is often deteriorated after an acute exacerbation, comorbidities, and lack of motivation are all considered as reasons for patients to decline PR. Other organizational factors and low rates of referrals are also important barriers [28-30].
In fact, we faced these challenges in our trial, it was difficult to recruit participants, a lot of patients did not wish to have a management after their exacerbation due to their poor health status. So, it was crucial to motivate the recruited patients to start and continue the rehabilitation program.
LIMITATION OF THE STUDY
Since it was impossible to blind the patients to the intervention, we cannot exclude a placebo effect as a possible mechanism participating in the observed improvements. However, we documented significant improvement in exercise performance, exercise duration, oxygen consumption, and dyspnea perception.
The study investigators were not also blinded to PR allocation because they were directly involved in the delivery of PR, and this cannot exclude an element of bias in the results of some questionnaires, but the cardiopulmonary test completed by each patient is highly standardized and was not subject to any possible bias.
CONCLUSION
EPR, after hospital discharge of COPD exacerbations, was associated with clinically significant improvement in the sensory perceptual, affective, and impact domains of dyspnea, and improvement in exercise performance and endurance. These findings support and recommend the implementation of EPR after an acute exacerbation of COPD.
DECLARATIONS
Availability of Data and Materials: The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate: Ethics approval from Ain Shams University, Faculty of Medicine research ethics committee (FWA00017585).Written informed consent signed by each patient to participate in the study. Consent obtained from each participant to publish his data collected in the study.
Competing Interests: None.
Acknowledgments: The investigators would like to thank AstraZeneca Company for their help and support to perform this research.
Funding: This study was funded by AstraZeneca.
Consent to Publish: Not applicable
Author Contribution: Amr Mounir Shoukri participated in the study design, collected the patient data, participated in the data analysis, and performed the clinical cardiopulmonary exercise tests. Ashraf Mokhtar Madkour participated in the study design, and reviewed the collected data. Tarek Mohamed Safwat participated in the study design and reviewed the collected data.
All authors read and approved the final manuscript.
REFERENCES
- Laveneziana P, Guenette JA, Webb KA, O'Donnell DE (2012) New physiological insights into dyspnea and exercise intolerance in chronic obstructive pulmonary disease patients. Expert Rev Respir Med 6: 651-662.
- Pitta F, Troosters T, Probst VS, Spruit MA, Decramer M, et al. (2006) Physical activity and hospitalization for exacerbation of COPD. Chest 129: 536-544.
- Man WD, Polkey MI, Donaldson N, Gray BJ, Moxham J (2004) Community pulmonary rehabilitation after hospitalisation for acute exacerbations of chronic obstructive pulmonary disease: randomised controlled study. BMJ 329: 1209.
- Casaburi R, ZuWallack R (2009) Pulmonary rehabilitation for management of chronic obstructive pulmonary disease. N Engl J Med 360: 1329-1335.
- Porszasz J, Emtner M, Goto S, Somfay A, Whipp BJ, et al. (2005) Exercise training decreases ventilatory requirements and exercise-induced hyperinflation at submaximal intensities in patients with COPD. Chest 128: 2025-2034.
- Laveneziana P, Parker CM, O'Donnell DE (2007) Ventilatory constraints and dyspnea during exercise in chronic obstructive pulmonary disease. Appl Physiol Nutr Metab 32: 1225-1238.
- O'Donnell DE, Laveneziana P (2007) Dyspnea and activity limitation in COPD: mechanical factors. COPD 4: 225-236.
- O’Donnell DE, Hamilton AL, Webb KA (2006) Sensory-mechanical relationships during high-intensity, constant-work-rate exercise in COPD. J Appl Physiol 101: 1025-1035.
- O'Donnell DE, Ora J, Webb KA, Laveneziana P, Jensen D (2009) Mechanisms of activity-related dyspnea in pulmonary diseases. Respir Physiol Neurobiol 167: 116-132.
- GOLD (2017) Global Strategy for the Diagnosis, Management and Prevention of COPD.
- Nici L, Raskin J, Rochester CL, Bourbeau JC, Carlin BW, et al. (2009) Pulmonary rehabilitation: What we know and what we need to know. J Cardiopulm Rehabil Prev 29: 141-151.
- Laveneziana P, O'Donnell DE, Ofir D, Agostoni P, Padeletti L, et al. (2009) Effect of biventricular pacing on ventilatory and perceptual responses to exercise in patients with stable chronic heart failure. J Appl Physiol 106: 1574-1583.
- Laveneziana P, Palange P, Ora J, Martolini D, O'Donnell DE (2009) Bronchodilator effect on ventilatory, pulmonary gas exchange, and heart rate kinetics during high-intensity exercise in COPD. Euro J App Physiol 107: 633-643.
- Borg GAV (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14: 377-381.
- Yorke J, Moosavi SH, Shuldham C, Jones PW (2010) Quantification of dyspnoea using descriptors: development and initial testing of the Dyspnoea 12. Thorax 65:21-26.
- Jones PW, Harding G, Berry P, Wiklund I, Chen WH, et al. (2009) Development and first validation of the COPD Assessment Test. Eur Respir J 34:648-654.
- Ofir D, Laveneziana P, Webb KA, Lam YM, O'Donnell DE (2008) Abnormal Ventilatory Responses to Incremental Cycle Exercise in Mild COPD. Am J Respir Crit Care Med 77: 622-629.
- O’Donnell DE, Laveneziana P, Ora J, Webb KA, Lam YM, et al. (2009) Evaluation of acute bronchodilator reversibility in patients with symptoms of GOLD stage I COPD. Thorax 64: 216-223.
- Ries AL, Bauldoff GS, Carlin BW, Casaburi R, Emery CF, et al. (2007) Pulmonary rehabilitation: joint ACCP/AACVPR evidence-based clinical practice guidelines. Chest 131: 4S-42S.
- Wilt TJ, Niewoehner D, MacDonald R, Kane RL (2007) Management of stable chronic obstructive pulmonary disease: a systematic review for a clinical practice guideline. Ann Intern Med 147: 639-653.
- Qaseem A, Snow V, Shekelle P, Sherif K, Wilt TJ, et al. (2007) Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline from the American College of Physicians. Ann Intern Med 147: 633-638.
- Lacasse Y, Goldstein R, Lasserson TJ, Martin S (2006) Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev :CD003793.
- Coventry PA, Hind D (2007) Comprehensive pulmonary rehabilitation for anxiety and depression in adults with chronic obstructive pulmonary disease: systematic review and meta-analysis. J Psychosom Res 63: 551-565.
- Nici L, Donner C, Wouters E, Zuwallack R, Ambrosino N, et al. (2006) American Thoracic Society/European Respiratory Society statement on pulmonary rehabilitation. Am J Respir Crit Care Med 173: 1390-1413.
- Seymour JM, Moore L, Jolley CJ, Ward K, Creasey J, et al. (2010) Outpatient pulmonary rehabilitation following acute exacerbations of COPD. Thorax 65:423-428.
- Greening NJ, Williams JE, Hussain SF, Harvey-Dunstan TC, Bankart MJ, et al. (2014) An early rehabilitation intervention to enhance recovery during hospital admission for an exacerbation of chronic respiratory disease: randomised controlled trial. BMJ 349: g4315.
- Ko FW, Dai DL, Ngai J, Tung A, Ng S, et al. (2011) Effect of early pulmonary rehabilitation on health care utilization and health status in patients hospitalized with acute exacerbations of COPD. Respirology 16: 617-24.
- Benzo R, Wetzstein M, Neuenfeldt P, McEvoy C (2015) Implementation of physical activity programs after COPD hospitalizations: lessons from a randomized study. Chron Respir Dis 12: 5-10.
- Fischer MJ, Scharloo M, Abbink JJ, Thijs-Van A, Rudolphus A, et al. (2007) Participation and drop-out in pulmonary rehabilitation: a qualitative analysis of the patient’s perspective. Clin Rehabil 21: 212-221.
- Arnold E, Bruton A, Ellis-Hill C (2006) Adherence to pulmonary rehabilitation: a qualitative study. Respir Med 100: 1716-1723.
Citation: Shoukri AM, Madkour AM, Safwat TM (2019) The Impact of Early Pulmonary Rehabilitation on the Multidimensional Aspects of Dyspnea and Exercise Performance Following Acute Exacerbation of Chronic Obstructive Pulmonary Disease: A Randomized Trial. J Pulm Med Respir Res 5: 031.
Copyright: © 2019 Amr Mounir Shoukri, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

